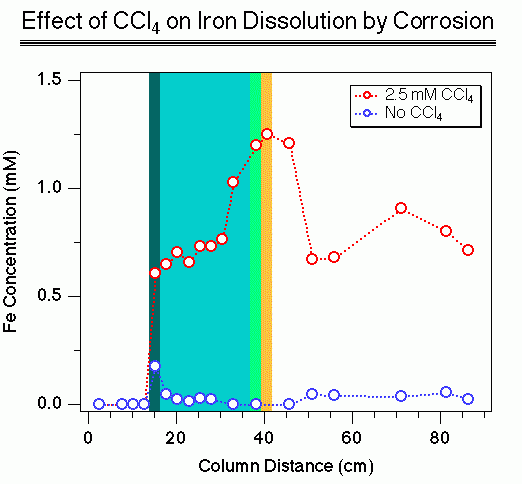
The figure represents an experiment performed to model geochemical effects that occur in an aquifer containing a "reactive barrier" of granular iron. Reactive barriers of this sort have recently emerged as one of the most promising new technologies for remediating contaminated groundwater.
The column contained a zone of sand, followed by a zone of granular iron, followed by another zone of sand. These zones are represented by the white, blue, and white regions of the figure. The three darkly colored areas represent local areas of precipitation caused by reaction of the water (that flowed from left to right) and the metal.
The data points show concentrations of dissolved iron measured in pore waters sampled from ports along the length of the column. The larger amount of iron released in the presence of carbon tetrachloride is a direct result of the reaction that results in its degradation.
A detailed description of the column experiment was published in Johnson, T. L., and P. G. Tratnyek. 1994. A column study of carbon tetrachloride dehalogenation by iron metal. Proceedings of the 33rd Hanford Symposium on Health & the Environment-In Situ Remediation: Scientific Basis for Current and Future Technologies. Pasco, WA. Vol. 2, pp. 931-947.
Submitted by Paul G. Tratnyek

Forum

Support
Gallery
Igor Pro 10
Learn More
Igor XOP Toolkit
Learn More
Igor NIDAQ Tools MX
Learn More





